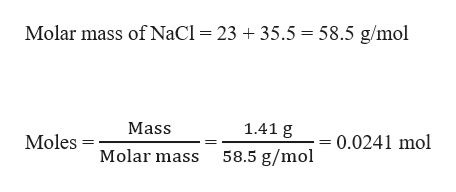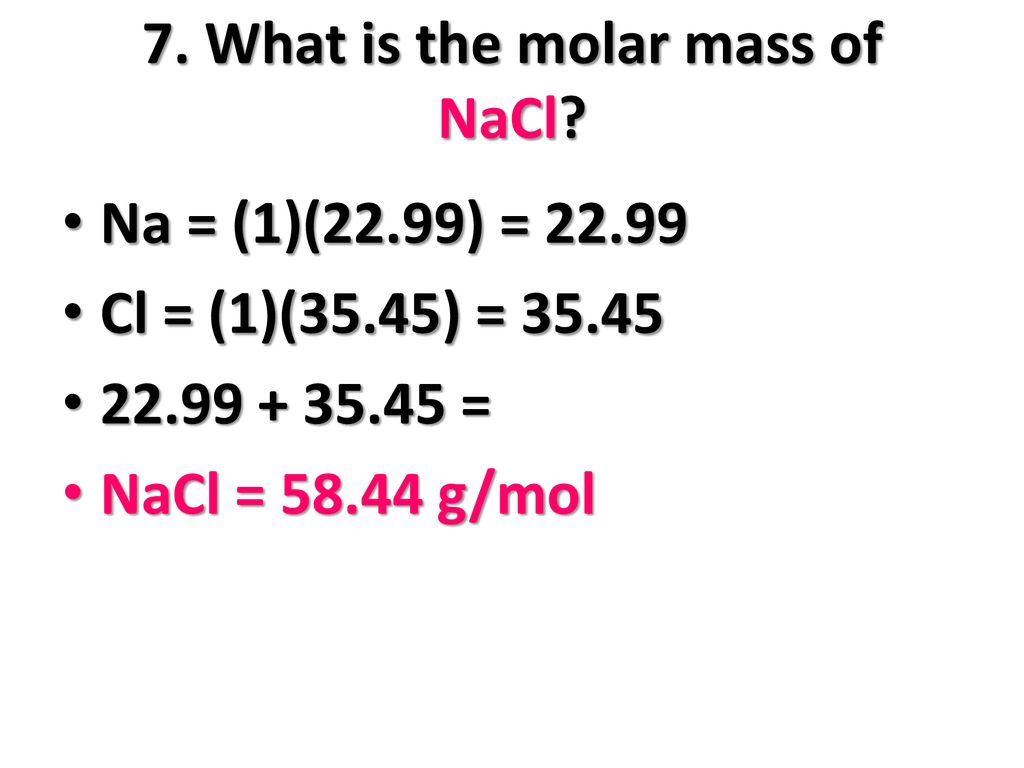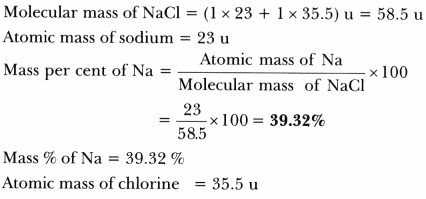
Calculate the mass per cent of each element of sodium chloride in one mole of it - CBSE Class 9 Science - Learn CBSE Forum

How Many NaCl Formula Units Does It Contain? express your answer using two significant figures - HomeworkLib
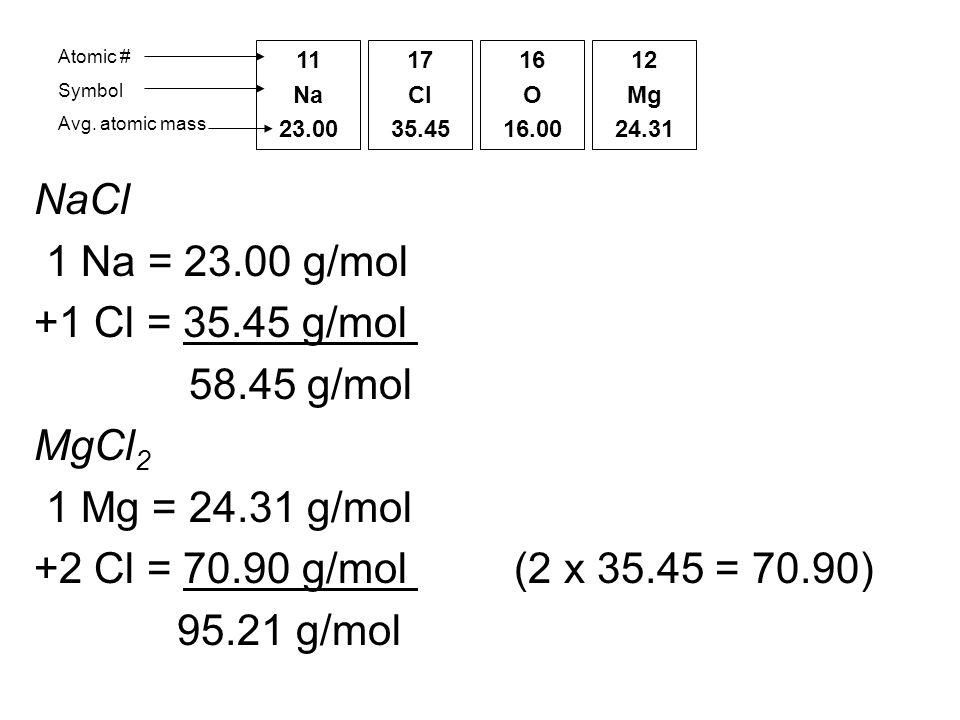
MOLAR MASS Molar mass of a substance = mass in grams of one mole of the substance. A compound's molar mass is NUMERICALLY equal to its formula mass. Formula. - ppt download

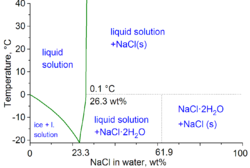
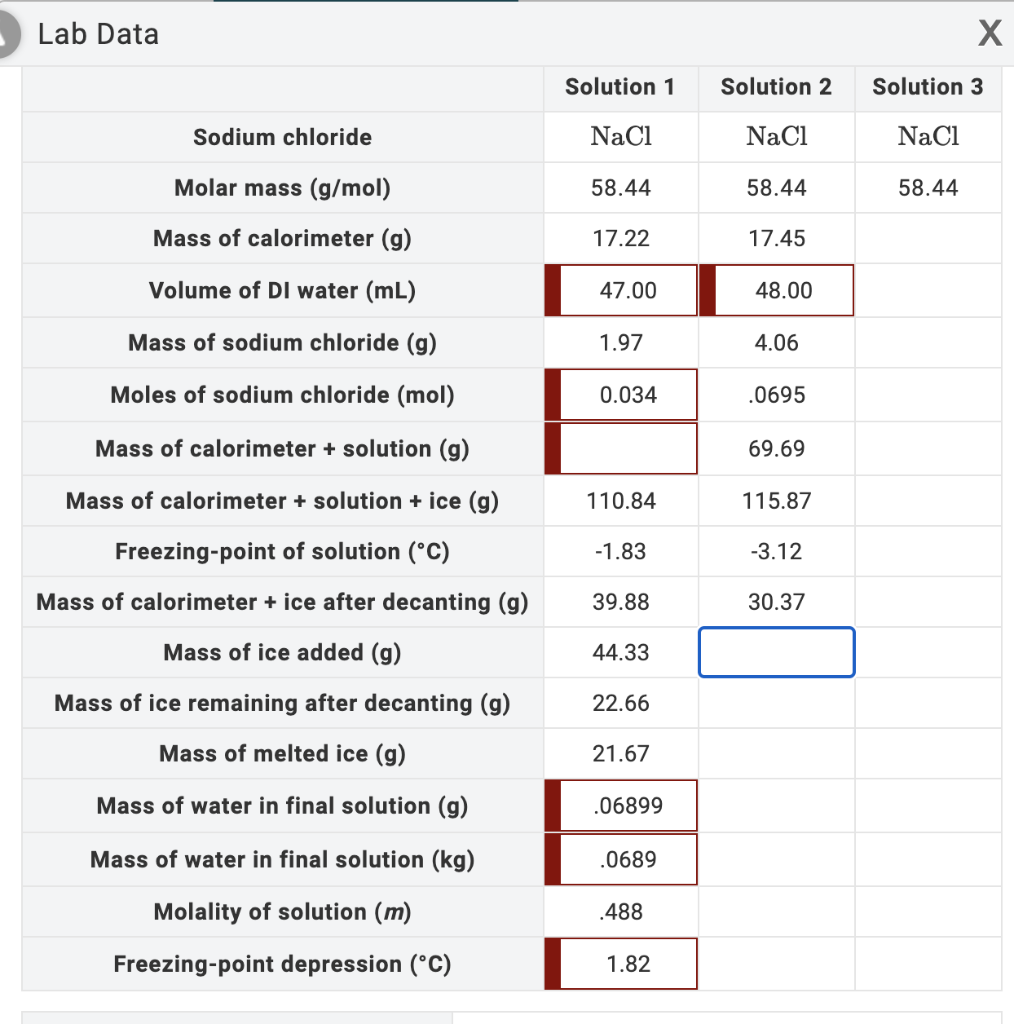
25. The molecular weight of NaCl determined by studying freezing point depression of its 0.5% aqueous solution is 30. The apparent degree of dissociation of NaCl is (1) 0.95 (2) 0.45 (3) 0.60 (4) 0.35

Dependence on the NaCl concentration of the apparent molar mass (a) and... | Download Scientific Diagram
Formula mass of NaCl is 58.45 g mol^-1 and density of its pure form is 2.167 g cm^-3. The average distance - Sarthaks eConnect | Largest Online Education Community

The reaction, 2A(g) + B(g) 3C(g) + D(g) , is begun with concentration of A and B both at initial value of 1 M . When equilibrium is reached, the concentration of